The Two Pillars of Equivalence
To get approved, a generic drug can't just "be similar." It has to prove two specific things: pharmaceutical equivalence and bioequivalence. Think of pharmaceutical equivalence as the "recipe" and bioequivalence as the "result."Pharmaceutical equivalence means the generic drug must have the exact same active ingredient, strength, dosage form (like a tablet or capsule), and route of administration (like oral or topical). It also has to be used for the same medical conditions as the original. While the active part must be identical, the "inactive" ingredients-like the dyes, fillers, or flavorings-can differ. This is why a generic pill might be blue while the brand is white, or why it might be round instead of oval.
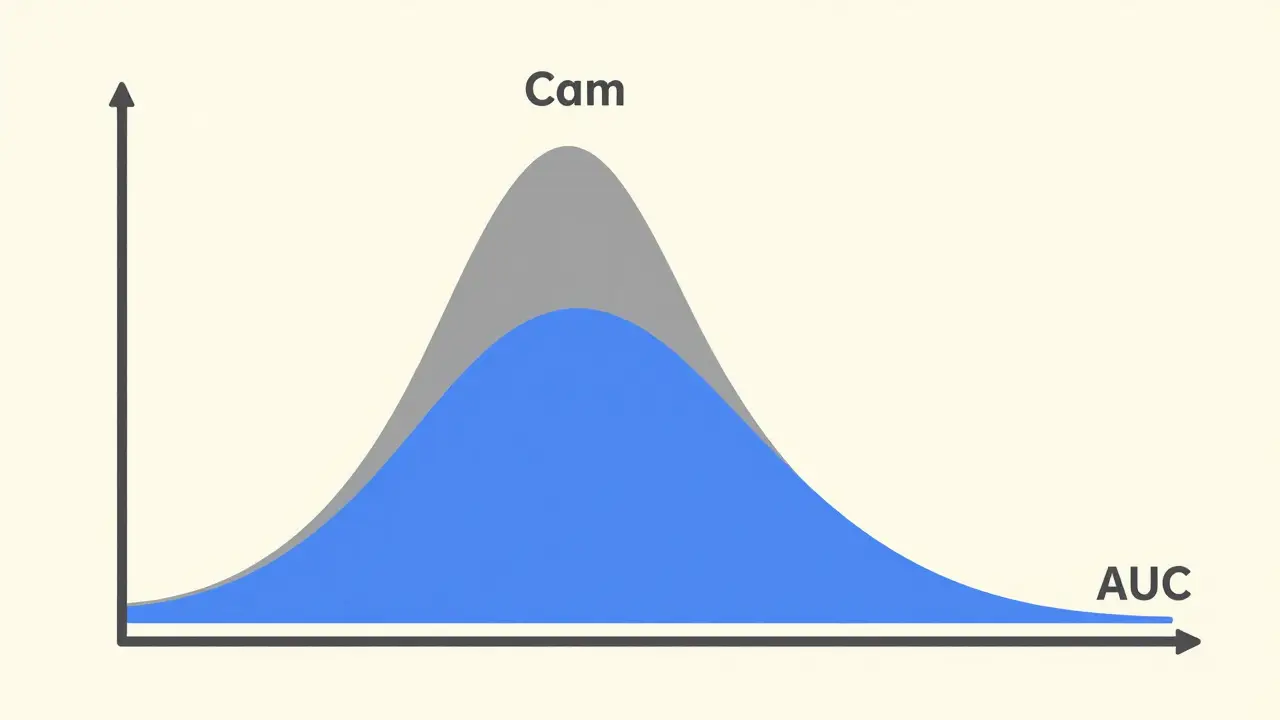
Bioequivalence is where the real science happens. The FDA doesn't want to redo the massive clinical trials the original company did to prove the drug works. Instead, the generic maker must prove that the drug reaches the bloodstream at the same rate and to the same extent as the brand name. They measure two main things: Cmax (the peak concentration of the drug in the blood) and AUC (the area under the curve, which represents the total drug exposure over time). To pass, the generic's performance must fall within a strict window of 80% to 125% of the brand-name's performance.
The ANDA Process: The Path to Approval
Unlike new drugs that require a massive New Drug Application (NDA), generics use a streamlined path called an Abbreviated New Drug Application (or ANDA). It's "abbreviated" because the manufacturer doesn't have to repeat animal or human safety trials-they just have to prove their version is the same as the one already approved.The ANDA process is a marathon, not a sprint. It often involves multiple review cycles and can take several months or even a year per cycle. The FDA's Office of Generic Drugs (OGD) works closely with the Office of Pharmaceutical Quality to tear apart the chemistry, manufacturing, and controls data. They aren't just checking the formula; they are checking the factory. If the manufacturing site isn't up to code, the drug doesn't get approved, no matter how good the lab results are.
| Requirement | Brand-Name (Innovator) | Generic (ANDA) |
|---|---|---|
| Active Ingredient | New/Original | Must be Identical |
| Clinical Trials | Full Safety & Efficacy | Bioequivalence Study |
| Manufacturing Standards | cGMP Compliant | cGMP Compliant |
| Inactive Ingredients | Proprietary | Can differ slightly |
| Approval Path | NDA (Full Application) | ANDA (Abbreviated) |
Manufacturing and the "Gold Standard" of Quality
Once the formula is approved, the actual making of the pill is governed by current Good Manufacturing Practices (known as cGMP). These aren't suggestions; they are federal regulations found in 21 CFR Parts 210 and 211.cGMPs ensure that every single batch of medicine is the same. This includes strict oversight of raw materials from the moment they enter the building to the moment the finished product is shipped. Companies must use validated testing methods to confirm the identity and purity of every lot. To keep everyone honest, the FDA conducts about 3,500 inspections of manufacturing facilities globally every year. Interestingly, about 50% of generics are actually made by the same big pharmaceutical companies that make the brand-name versions, often in the same factories.
Testing for Stability and Shelf Life
A drug that works on day one but fails by month six is a failure. This is why the FDA requires rigorous stability testing. Manufacturers must prove the drug stays potent and pure throughout its entire shelf life.They typically do this through two types of tests:
- Accelerated Stability Studies: Drugs are put in high-stress environments (like 40°C with 75% humidity) for six months to see how they break down.
- Long-term Studies: Drugs are kept at recommended storage temperatures for 12 to 24 months to mirror real-world use.
For most small-molecule drugs, a bioequivalence study involves 24 to 36 healthy volunteers who take a single dose of the drug. Their blood is monitored to ensure the generic hits the same peaks and troughs as the brand name. If the data deviates from that 80-125% window, the application is rejected.
The Challenge of Complex Generics
Not all drugs are simple tablets. Some are Complex Generics-think of inhalers, topical creams, or long-acting injectables. You can't just measure a blood level for an inhaler and call it a day; the drug has to reach the lungs in a very specific way to work.Because of this, the FDA has released over 2,100 product-specific guidances. These provide a blueprint for exactly how to test these tricky medications. Some require "Q3 sameness," which means the physical and chemical properties (like particle size or viscosity) must be nearly identical to the Reference Listed Drug (RLD). Even with these rules, complex generics are harder to get right. For example, about 17% of complex product categories require testing methods that go way beyond standard blood tests.
Real-World Performance and Patient Experience
For the vast majority of people, the switch to generics is seamless. A Consumer Reports survey showed that 89% of U.S. adults are satisfied with their generic meds. However, there is a small group of medications where the margins are incredibly thin.These are called narrow therapeutic index (NTI) drugs. In these cases, a tiny change in the amount of drug absorbed can be the difference between a dose that works and one that is toxic or ineffective. A study in JAMA Internal Medicine found that about 12.3% of patients switching between different generic versions of levothyroxine (a thyroid medication) experienced fluctuations in their thyroid levels. This doesn't mean the generics are "bad," but rather that different manufacturers' inactive ingredients can slightly alter how a person absorbs the drug.
Are generic drugs less effective than brand-name drugs?
No. By law, generic drugs must be bioequivalent to the brand-name drug, meaning they must deliver the same amount of active ingredient into the bloodstream at the same rate. The FDA ensures they have the same strength, purity, and stability.
Why do some generics look different from the brand name?
FDA regulations allow generic manufacturers to use different inactive ingredients, such as colors, fillers, and binders. As long as these ingredients don't interfere with how the drug works, the physical appearance of the pill can vary.
What is the "80% to 125%" rule?
This is the bioequivalence window. The 90% confidence interval for the ratio of the generic drug's peak concentration (Cmax) and total exposure (AUC) must fall between 80% and 125% of the brand-name drug's values to be considered equivalent.
What happens if a generic manufacturer fails an FDA inspection?
If a facility fails to meet current Good Manufacturing Practices (cGMP), the FDA can issue warning letters, reject pending ANDA applications, or in severe cases, halt production and order recalls of existing products.
Which drugs are the most "risky" to switch to generics?
While most generics are safe, Narrow Therapeutic Index (NTI) drugs-such as some seizure medications, blood thinners, and thyroid hormones-can be more sensitive. Some patients may experience slight differences when switching between different generic brands of these specific drugs.
